Associated Research Director
Associated Research Director
Research Infrastructures for Marine Biological Resources Department
Advanced Microscopy Center
Tel. +39 081 5833289 - +39 081 5833361
Fax: +39 081 7641355
e-mail luigia.santella(at)szn.it
Research Interests
Dr. Santella's research focuses on the structural and molecular mechanisms of signal transduction that induce physiological changes in oocytes and eggs during meiotic maturation and fertilization. Over the past two decades, she has primarily concentrated on the fluctuations of intracellular Ca2+ during these processes, using starfish and sea urchins as model organisms. Her research on immature and maturing starfish oocytes—ideal candidates for microinjection and imaging analysis—has established that the Ca²⁺ signals at fertilization can be triggered by the combined effects of Ca²⁺-linked second messengers, such as NAADP and InsP₃. These messengers play distinct roles in initiating and propagating the fertilization Ca²⁺ response, respectively. In contrast, cADPr may have a modulatory function. She has also demonstrated that certain Ca²⁺ signals can originate and propagate within specific subcellular domains, including the nucleus and subplasmalemmal regions. Recent studies have shown that the actin cytoskeleton of the egg cortex significantly affects the patterns of intracellular Ca2+ increases in starfish, and this finding has been applied to sea urchin eggs as well. Dr. Santella has been actively involved in promoting international collaboration and facilitating intellectual exchanges, in addition to her research efforts. She has organized several successful international symposia and is often invited to speak at conferences in Europe, the USA, and Asia. From 1993 to 2018, she made annual visits to the Asamushi Research Center for Marine Biology at Tohoku University in Japan to perform experiments on the local starfish species. Additionally, she has lectured in educational programs supported by EMBO and NSF in Brazil and Uruguay.
Appointments and Memberships
2022 Sigma Xi Honor Society Member
2020 Member of the Editorial Board of Cells
2015-2024 Editor of Biochemical Biophysical Research Communications
2015 Member of the Scientific Committee of the European Calcium Society
2014 Member of the Editorial Board of Zoological Letters
2014-2024 Member of the Editorial Board of Zygote
2004-2009 Member of the Editorial Board of Physiological Reviews
2004 Member of the Academia Europaea
Invited Speaker at International Meetings/Symposia/Workshops/Conferences such as FEBS, Ca2+-Binding Proteins, and Ca2+ Function in Health and Disease, EMBO, FASEB Research, Gordon, and Advanced Courses.
Organizer of International Symposia
2019. Santella L. Biochemical Biophysical Research Communications Editorial Meeting, Naples, Italy, 6-8 December.
2013. Santella L. and Chun JT. International Symposium on: The dynamically active egg: The legacy of Ernest Everett Just (held to celebrate the 130th anniversary of the birth of Ernest Everett Just), Naples, 13 May.
2009. Santella L, De Natale G, Baffioni C, Cavalli A. Academia Europaea 21st Annual Conference on Risks, Environment and Sustainable Development, Naples, Italy. 24—26 September.
2004. Santella L, Cattaneo E, Rizzuto R, Carafoli E. EMBO Workshop on Calcium Signalling and Disease, Capri, Italy.
1997. Santella L, Bachs O, Nicotera P, Carafoli E. First European Conference on Calcium Signalling in the Cell Nucleus, Baia Paraelios, Italy. Reports on the conference have appeared in Cell (92:17-23, 1998) and Cell Calcium (22:313-319, 1997).
Special Issues Organizer
Special Issue on The Cell Biology of Fertilization (2023). In: Cells, Guest Editors: Santella L, Limatola N.
https://www.mdpi.com/journal/cells/special_issues/0W5YL3TJIE
Special Issue on Waves in Fertilization, Cell Division, and Embryogenesis (2021). In: Biosystems, Guest Editors: Santella L, Gordon R, Chen Z, Tuszynski J.
https://www.sciencedirect.com/journal/biosystems/special-issue/1022D42HXMT
Special Issue on Cytoskeletal Structure and Dynamics in Fertilization and Early Development (2019). In: Frontiers in Cell and Developmental Biology, Guest Editors: Santella L, Chun JT.
https://www.frontiersin.org/research-topics/23330/cytoskeletal-structure-and-dynamics-in-fertilizationand-early-development
Special Issue on Actin Cytoskeleton Dynamics (2018). In: Biochemical Biophysical Research Communications. Guest Editors, Santella L, Mabuchi I. 506, 307-422.
Special issue on Fertilization and Early Development (2013). In: Biochemical Biophysical Research Communications. Guest Editors, Lennarz WJ, Santella L. 450, 1133-1230.
Publications (h-index: 39)
Selected Publications
Limatola N, Pirozzi M, Caramiello D, Chun JT, Santella L. (2025) The Binding of Concanavalin A to the Surface of Intact and Denuded Sea Urchin Eggs Affects the Fertilization Process by Altering the Structural Dynamics of Actin Filaments. Cells, 14(23), 1867; https://doi.org/10.3390/cells14231867
Limatola N, Chun JT, Schmitt JL, Lehn JM, Santella L. The Effect of Synthetic Polyamine BPA-C8 on the Fertilization Process of Intact and Denuded Sea Urchin Eggs. (2024) Cells, 13(17), 1477; https://doi.org/10.3390/cells13171477
Mangini M, Limatola N, Ferrara MA, Coppola G, Chun JT, De Luca AC. Santella L. (2024) Application of Raman spectroscopy to the evaluation of F-actin changes in sea urchin eggs at fertilization. Zygote, 32, 38-48; https://doi.org/10.1017/S0967199423000552
Limatola N, Chun JT, Chiba K, Santella L. (2023) Dithiothreitol Affects the Fertilization Response in Immature and Maturing Starfish Oocytes. Biomolecules, 13(11), 1659; https://doi.org/10.3390/biom13111659
Limatola N, Chun JT, Schneider SC, Schmitt JL, Lehn JM, Santella L. (2023) The Effect of Acidic and Alkaline Seawater on the F-Actin-Dependent Ca2+ Signals Following Insemination of Immature Starfish Oocytes and Mature Eggs. Cells, 12(5), 740; https://doi.org/10.3390/cells12050740
Santella L, Chun JT. (2022) Structural actin dynamics during oocyte maturation and Fertilization. Biochem Biophys Res Commun 633, 13-16; https://www.sciencedirect.com/science/article/pii/S0006291X22012438
Limatola N, Chun JT, Santella L. (2022) Species-Specific Gamete Interaction during Sea Urchin Fertilization: Roles of the Egg Jelly and Vitelline Layer. Cells, 11(19), 2984; https://doi.org/10.3390/cells11192984
Limatola N, Chun JT, Santella L. (2022) Regulation of the Actin Cytoskeleton-Linked Ca2+ Signaling by Intracellular pH in Fertilized Eggs of Sea Urchin. Cells, 11(9), 1496; https://doi.org/10.3390/cells11091496
Limatola N, Chun JT, Cherraben S, Schmitt JL, Lehn JM, Santella L. (2021) Effects of dithiothreitol on Fertilization and early development in sea urchin. Cells, 10(12), 573; https://doi.org/10.3390/cells10123573
Limatola N, Vasilev F, Santella L, Chun JT. (2020) Nicotine induces polyspermy in sea urchin eggs through a non-cholinergic pathway modulating actin dynamics. Cells , 9(1), 63; https://doi.org/10.3390/cells9010063
Santella L, Limatola N, Chun JT. (2020) Cellular and molecular aspects of oocyte maturation and Fertilization: a perspective from the actin cytoskeleton. Zool Lett., 6, 1-21; https://link.springer.com/article/10.1186/s40851-020-00157-5
Limatola N, Chun JT & Santella L. (2020) Effects of salinity and pH of seawater on the reproduction of the sea urchin Paracentrotus lividus. Biol Bull., 239, 13-23.
Limatola N, Vasilev F, Chun JT & Santella L. (2019) Sodium-mediated fast electrical depolarization does not prevent polyspermic Fertilization in Paracentrotus lividus eggs. Zygote, 27, 241-24.
Vasilev F, Limatola N, Chun JT & Santella L. (2019) Contributions of suboolemmal acidic vesicles and microvilli to the intracellular Ca2+ increase in the sea urchin eggs at Fertilization. Int J Biol Sci., 15, 757; https://www.ijbs.com/v15p0757.htm
Limatola N, Vasilev F, Chun JT & Santella L. (2019) Altered actin cytoskeleton in ageing eggs of starfish affects fertilization process. Exp Cell Res., 381, 179-190.
Limatola N, Chun JT, Kyozuka K, Santella L. (2015) Novel Ca2+ increases in the maturing oocytes of starfish during the germinal vesicle breakdown. Cell Calcium, 58, 500-510.
Chun JT, Limatola N, Vasilev F, Santella L. (2014) Early events of Fertilization in sea urchin eggs are sensitive to actin-binding organic molecules. Biochem Biophys Res Commun., 450, 1166-74.
Vasilev F, Chun JT, Gragnaniello G, Garante E, Santella L. (2012). Effects of ionomycin on egg activation and early development in starfish. PLoS One, 7, e39231; https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0039231
Chun JT, Puppo A, Vasilev F, Gragnaniello G, Garante E, Santella L. (2010) The biphasic increase of PIP2 in the fertilized eggs of starfish: new roles in actin polymerization and Ca2+ signaling. PLoS One, 5, e14100; https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0014100
Puppo A, Chun JT, Gragnaniello G, Garante E, Santella L. (2008). Alteration of the cortical actin cytoskeleton deregulates Ca2+ signaling, monospermic Fertilization, and sperm entry. PLoS One 3, e3588; https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0003588
Kyozuka K, Chun JT, Puppo A, Gragnaniello G, Garante E, Santella L. (2008). Actin cytoskeleton modulates calcium signaling during maturation of starfish oocytes. Dev Biol., 320, 426-35.
Lim D, Ercolano E, Kyozuka K, Nusco GA, Moccia F, Lange K, Santella L. (2003) The M-phase-promoting factor modulates the sensitivity of the Ca2+ stores to inositol 1, 4, 5-trisphosphate via the actin cytoskeleton. J Biol Chem., 278, 42505-42514; https://www.jbc.org/article/S0021-9258(20)82794-X/fulltext
Lim D, Lange K, Santella L. (2002) Activation of oocytes by latrunculin A. The FASEB J., 16, 1050-1056.
Lim D, Kyozuka K, Gragnaniello G, Carafoli E, Santella L. (2001) NAADP+ initiates the Ca2+ response during Fertilization of starfish oocytes. The FASEB J., 15, 2257-226.
Santella L. , Kyozuka K. (1994) Reinitiation of meiosis in starfish oocytes requires an increase in nuclear Ca2+. Biochem Biophys Res Commun., 203, 674-680.
Websites or online articles
2015 Small World in Motion Competitionn (Video)
https://atlasofscience.org/the-fertilization-process-a-new-way-to-look-at-an-old-phenomenon/
International Symposium - The dynamically active egg: The legacy of Ernest Everett Just
Everett_Just_at_the_Stazione_Zoologica_in_Naples_Celebration_of_a_Friendship
Photos

Fluorescent labeling of live Paracentrotus lividus sperm and egg envelopes. (A) Fresh sperm were diluted in natural seawater containing polyamine BPA-C8-Cy3 (green) and Hoechst-33342 (blue). An intact egg was labeled with Alexa Fluor 633 Con A and viewed in a non-confocal microscopy mode, allowing visualization of the jelly coat (JC). (C) Two denuded eggs labeled with Alexa Fluor 633 Con A and viewed in non-confocal microscopy mode.

Transmission and scanning electron microscopy of early surface changes during fertilization of sea urchin eggs

Sperm-egg fusion beneath the fertilization envelope following insemination of a sea urchin egg.
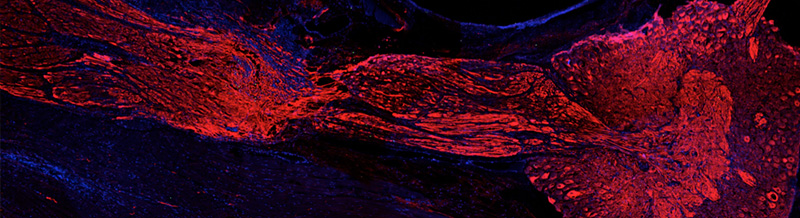
F-actin staining in a starfish oocyte microinjected with Alexa Fluor-568 conjugated phalloidin.
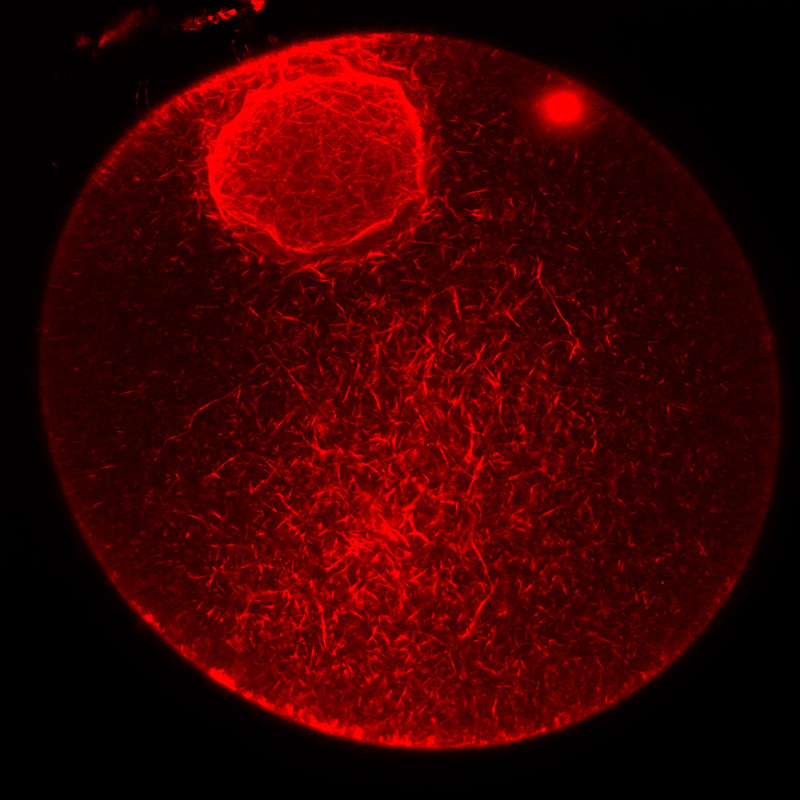
F-actin staining in the nucleus and cytoplasm of a starfish immature oocyte.

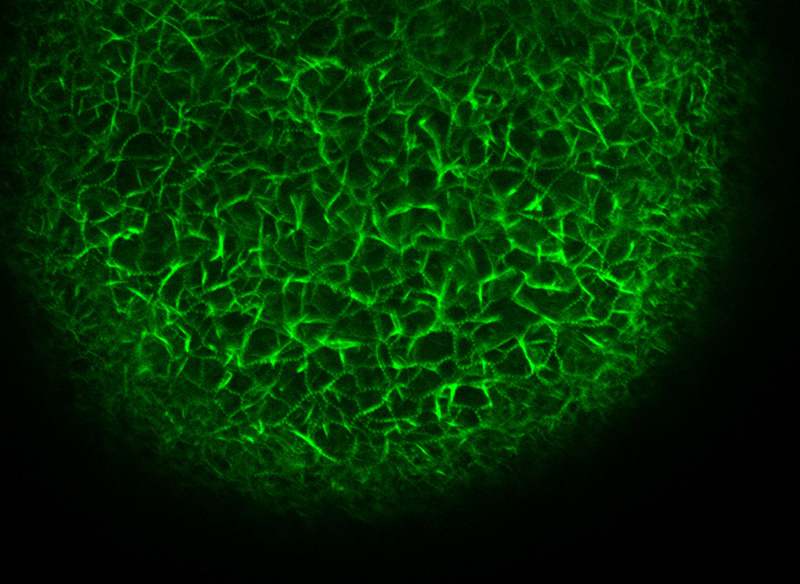
F-actin spikes into the perivitelline space of a starfish egg following uncaging of injected InsP3 visualized by labelling PIP2 with the RFP-PH domain of the PLC1.
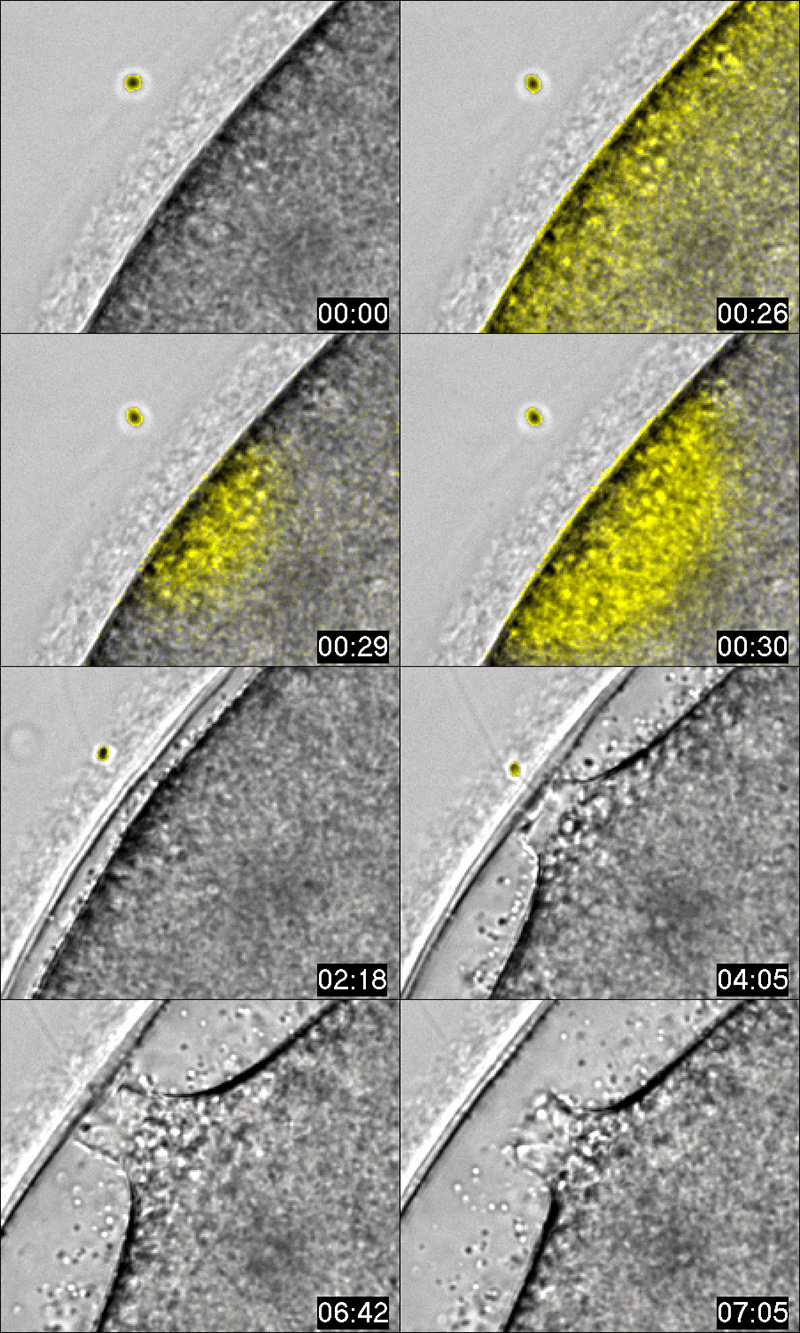
The interaction of the acrosomal process, visible at 04:05 min, of the sperm (yellow circle) with the jelly coat of a starfish egg triggers intracellular Ca2+ increases (yellow fluorescence). Detailed view of the sperm entry site during fertilization.

Sperm incorporation into a starfish egg by cytoplasmic protrusions crossing the fertilization envelope.